Surface Tension Examples, Causes & Formula | What is Surface Tension in Physics? - Video & Lesson Transcript | Study.com

Equilibrated Interfacial Tension Data of the CO2–Water System at High Pressures and Moderate Temperatures | Journal of Chemical & Engineering Data

A capillary tube of radius 'r' is immersed in water and water rises in to a height 'h' . Mass of water in the capillary tube is 5 × 10^-3 kg .

Applied Sciences | Free Full-Text | Effect of Hydrogen Bonding on the Surface Tension Properties of Binary Mixture (Acetone-Water) by Raman Spectroscopy

Table 1 from Surface Tension of Supercooled Water: No Inflection Point down to -25 °C. | Semantic Scholar
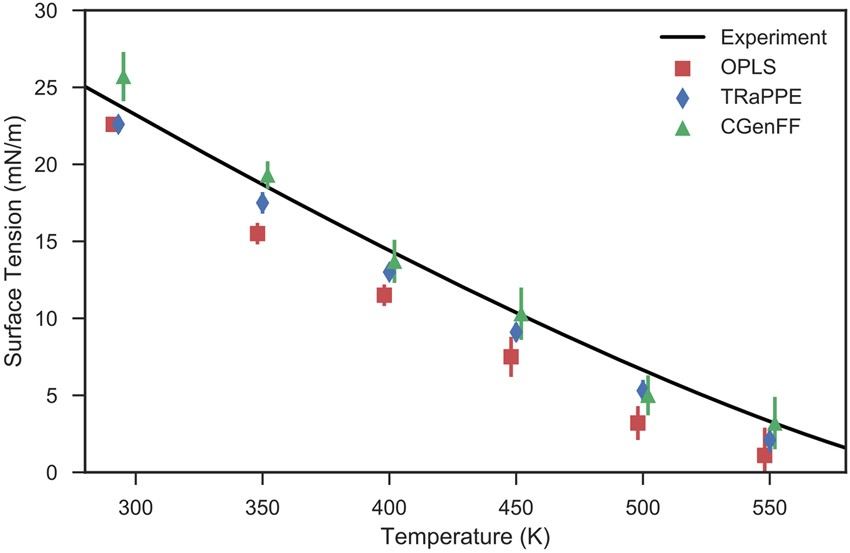
The Water-Alkane Interface at Various NaCl Salt Concentrations: A Molecular Dynamics Study of the Readily Available Force Fields | Scientific Reports

SOLVED: The surface tension of water is 72 mN/m at 25 degrees celsius. The decane has a surface tension of 24mN/m. The interfacial tension of the two liquids is 51 mN/m. Estimate
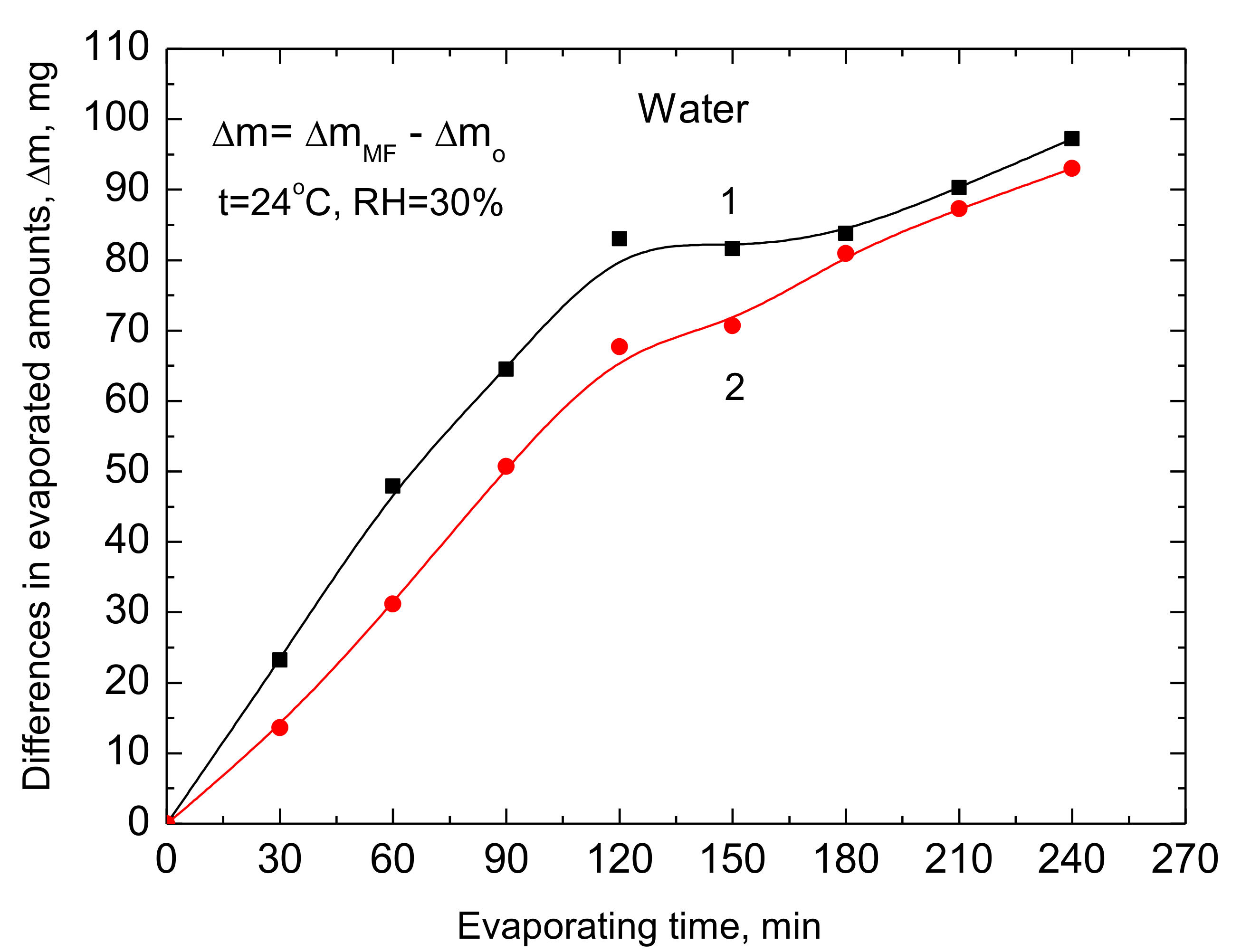
Colloids and Interfaces | Free Full-Text | Influence of Magnetic Field on Evaporation Rate and Surface Tension of Water

The height of water in a capillary tube of radius 2 cm is 4 cm . What should be the radius of capillary, if the water rises to 8 cm in tube?

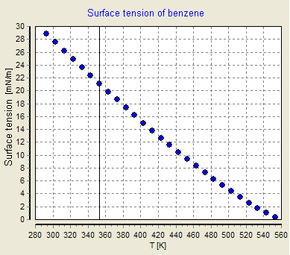





![PDF] Surface Tension Determination with a Teflon Rod | Semantic Scholar PDF] Surface Tension Determination with a Teflon Rod | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f5cd29f13e247575fa22038eafc383a040670fa1/3-Table1-1.png)