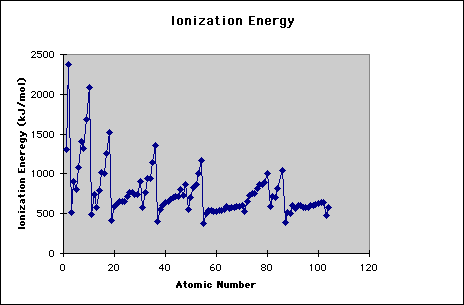
First and second ionisation energies of magnesium are `5.7` and `15.035eV` respectively. The amount - YouTube
The correct order of decreasing second ionization energy of Li, Be, Ne,(1) Ne>B>Li>C>BeC, B(2) Li>Ne C>B>Be(3) Ne>C>B>Be>Li4)
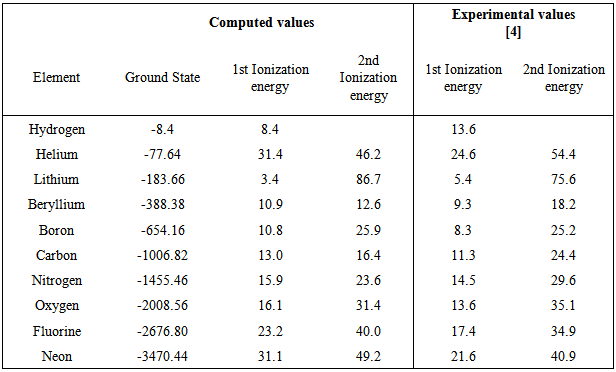
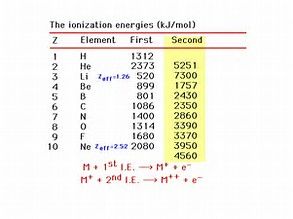
Computation of the First and Second Ionization Energies of the First Ten Elements of the Periodic Table Using a Modified Hartree-Fock Approximation Code
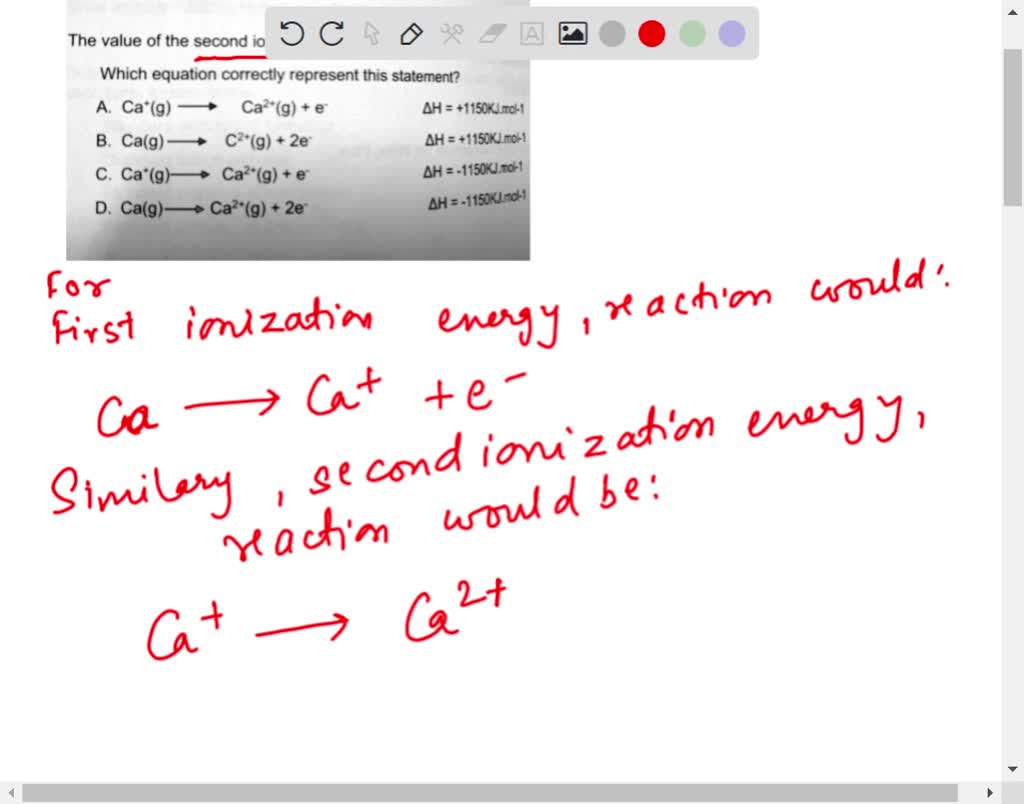
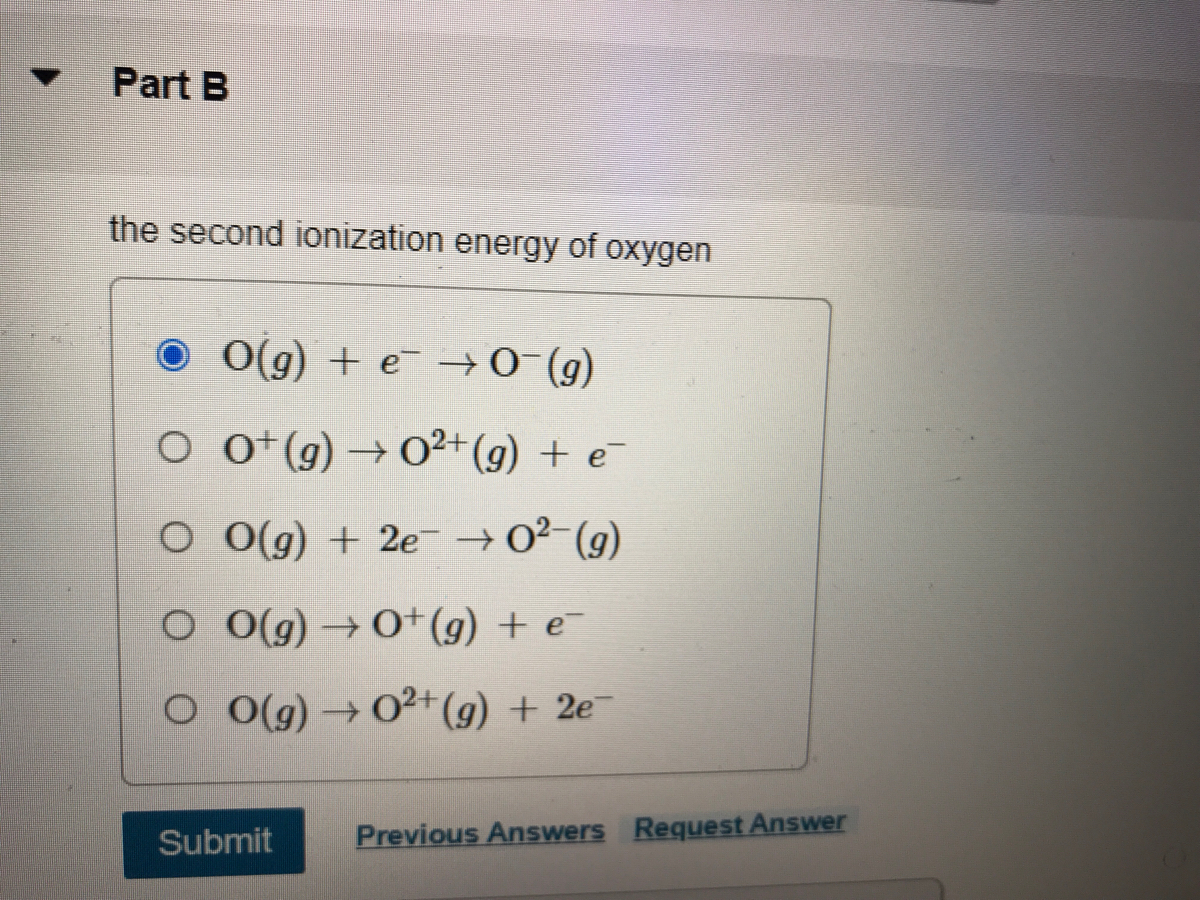
SOLVED: The value of the second ionisation energy of calcium is 1150 Klmol1 Which equation correctly represent this statement? A Ca (g) Ca?*(g) + e AH = +1150KJ,mol:1 B. Ca(g) c*(g) -
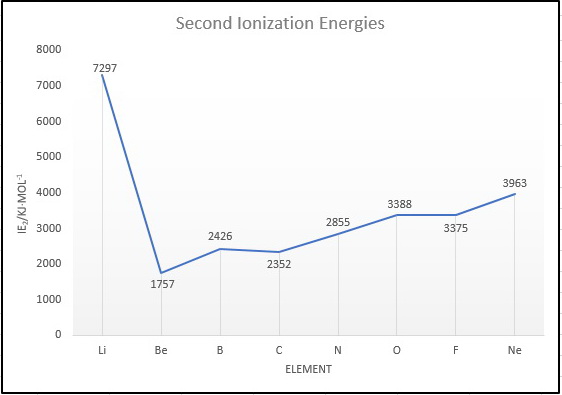
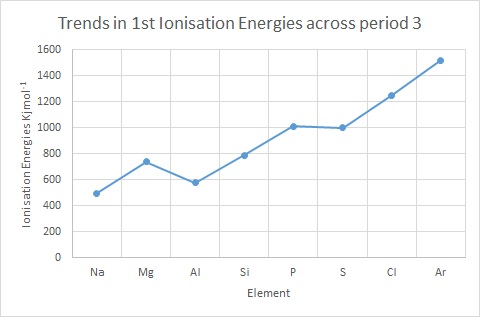
What is the order of the second ionization energies for elements in the second row of the Periodic Table? | Socratic

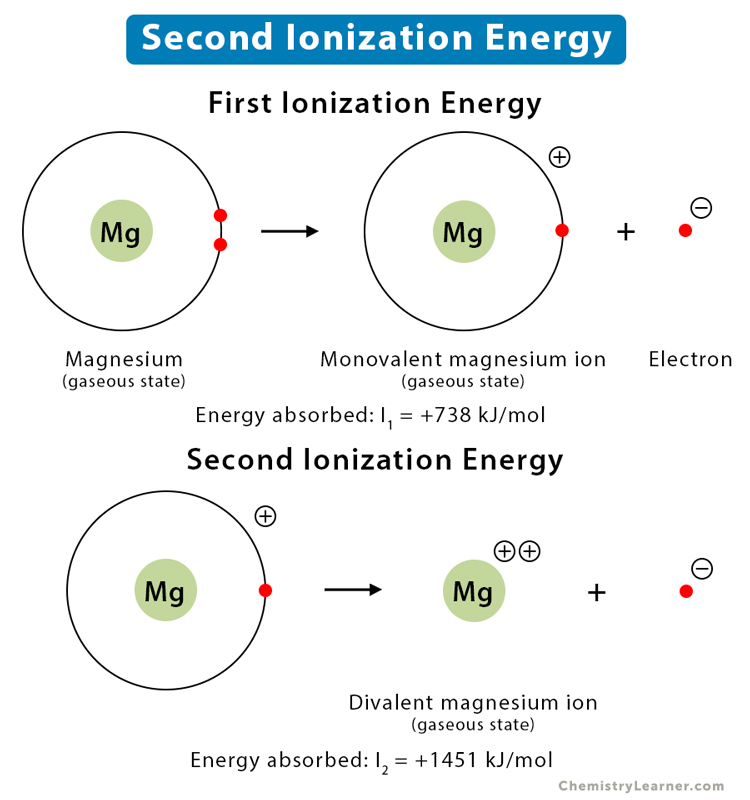
Difference Between First and Second Ionization Energy (I1E vs I2E) | Compare the Difference Between Similar Terms
The correct order of decreasing second ionization energy of Li, Be, Ne,(1) Ne>B>Li>C>BeC, B(2) Li>Ne C>B>Be(3) Ne>C>B>Be>Li4)
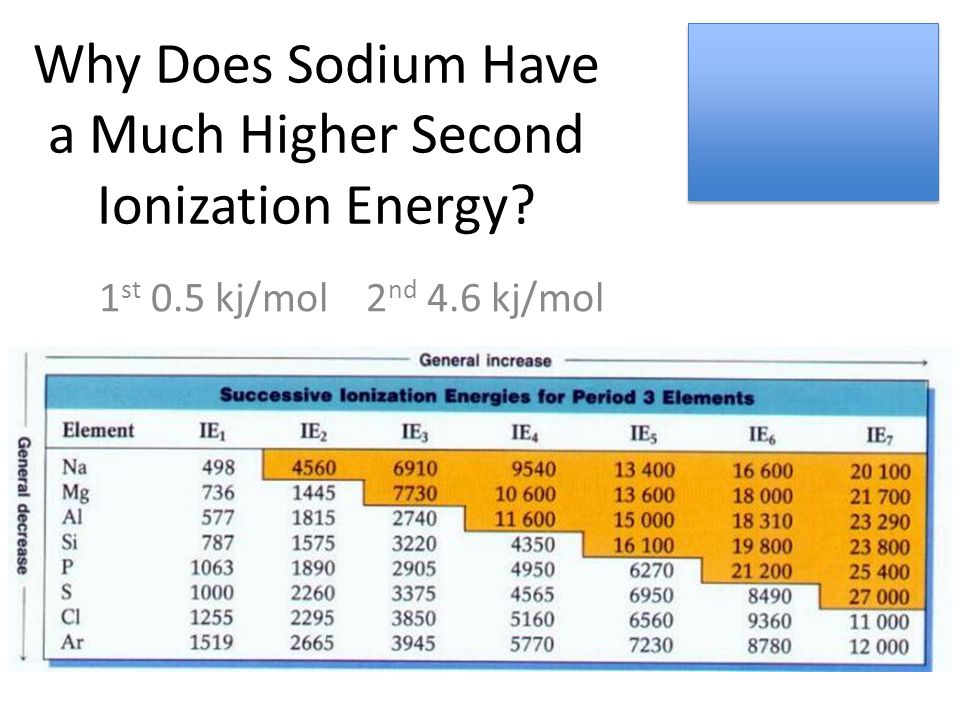
Why Does Sodium Have a Much Higher Second Ionization Energy? 1 st 0.5 kj/mol 2 nd 4.6 kj/mol. - ppt download