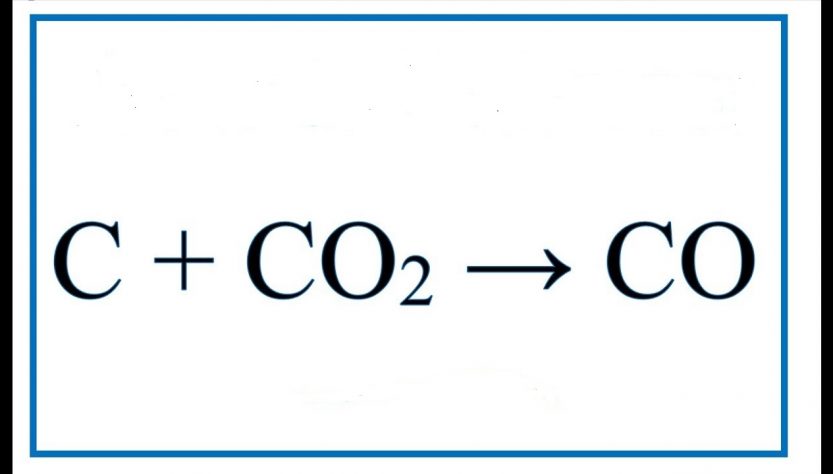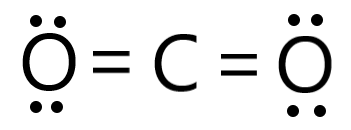Rasprodaja Detektor Kvalitete Zraka Zidni Monitor CO2 Mjerač Temperature I Vlage U Prostoru Detektor Kvalitete Zraka CO2 Za Kućni Ured C \ Mjernih i Analitičkih instrumenata ~ Masazerzalice.com.hr
22g of CO2 at 27 celsius is mixed in a closed container with 16 gram of O2 at 37 Celsius if both gases are considered as Ideal kinetic theory of gases then
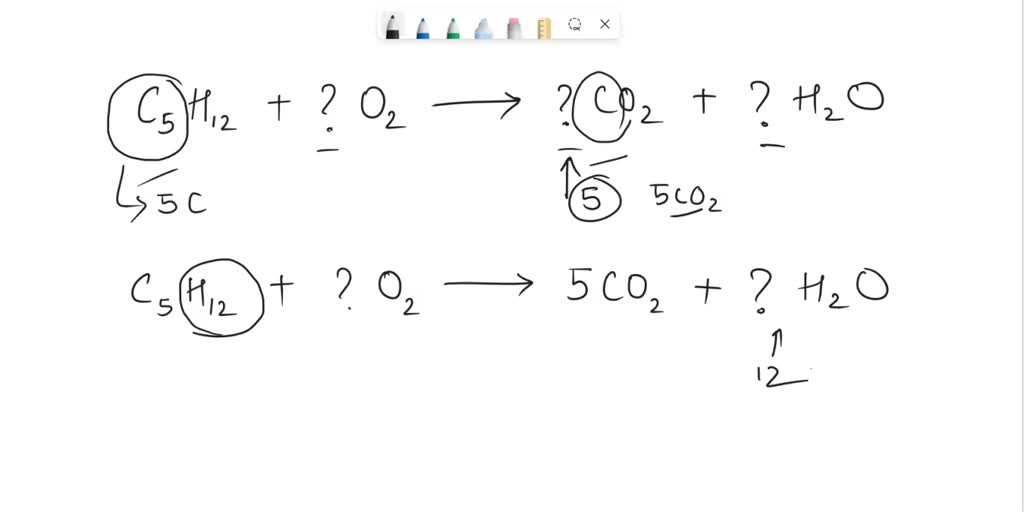
SOLVED: What is the coefficient for carbon dioxide in the balanced equation? C5H12 + ? O2 → ? CO2 + ? H2O A) 2 B) 4 C) 5 D) 6 E) 8

Atomically Dispersed Indium‐Copper Dual‐Metal Active Sites Promoting C−C Coupling for CO2 Photoreduction to Ethanol - Shi - 2022 - Angewandte Chemie International Edition - Wiley Online Library

For the reaction, C (s) + CO2 (g) 2CO (g) , the partial pressures of CO2 and CO are 2.0 and 4.0 atm respectively at equilibrium. The Kp for the reaction is:

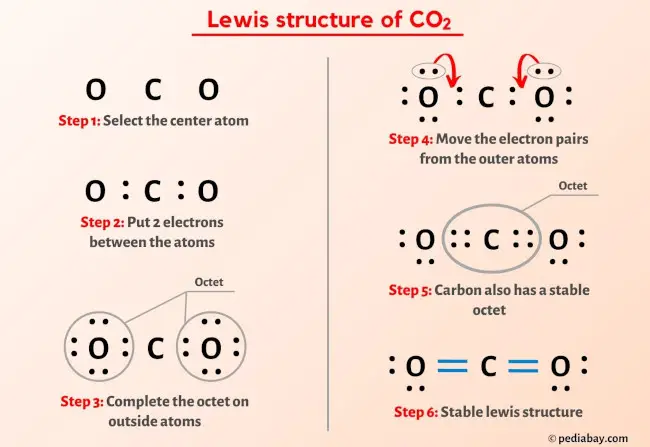
Consider the following reactions, C (s) + O2 (g) → CO2 (g), Δ H = - 94 kcal 2CO (g) + O2 → 2 CO2 (g) , Δ H = - 135.2 kcalThen, the heat of formation of CO (g) is:
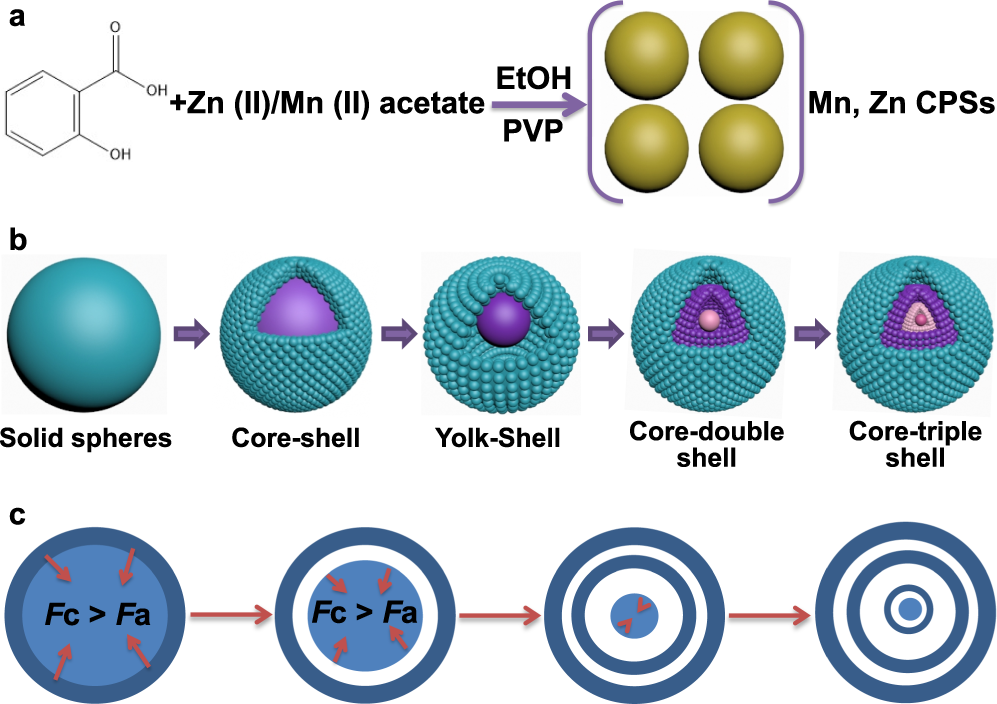
Sustained CO2-photoreduction activity and high selectivity over Mn, C-codoped ZnO core-triple shell hollow spheres | Nature Communications