Falsified Medicines Directive - Legal framework and Implementation - Pharma Serialization, Aggregation and Track and Trace Software by SoftGroup

Book Excerpt: Fundamentals of EU Regulatory Affairs, Seventh Edition, Chapter 8: European Union Fals | RAPS

Final aspect of the EU Falsified Medicines Directive to be Implemented by February 2019 - PharSafer® - Specialists in Global Clinical and Post Marketing Drug Safety
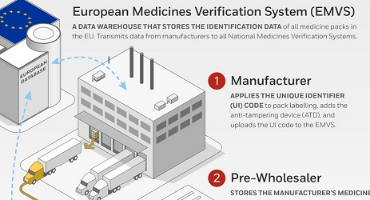
How does the Falsified Medicines Directive (FMD) EU directive supply chain work ? | Barcode Technoloiges Ltd / Barcode - UK