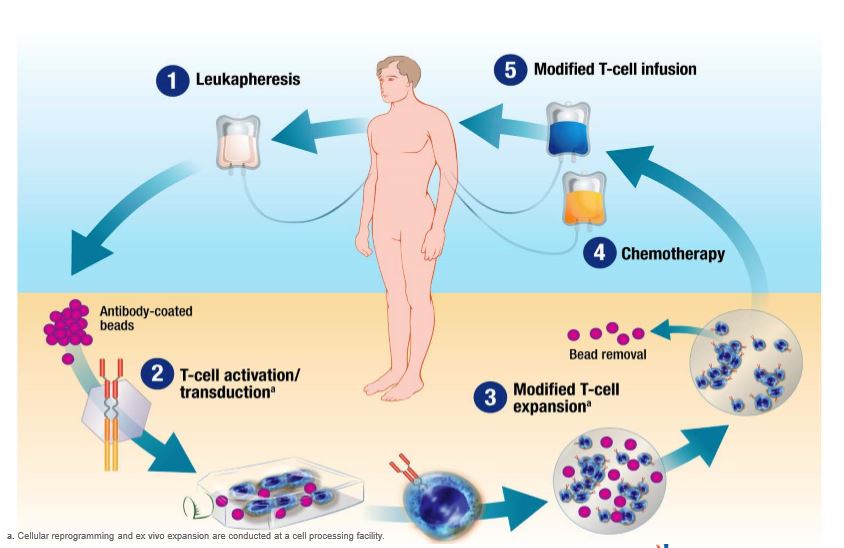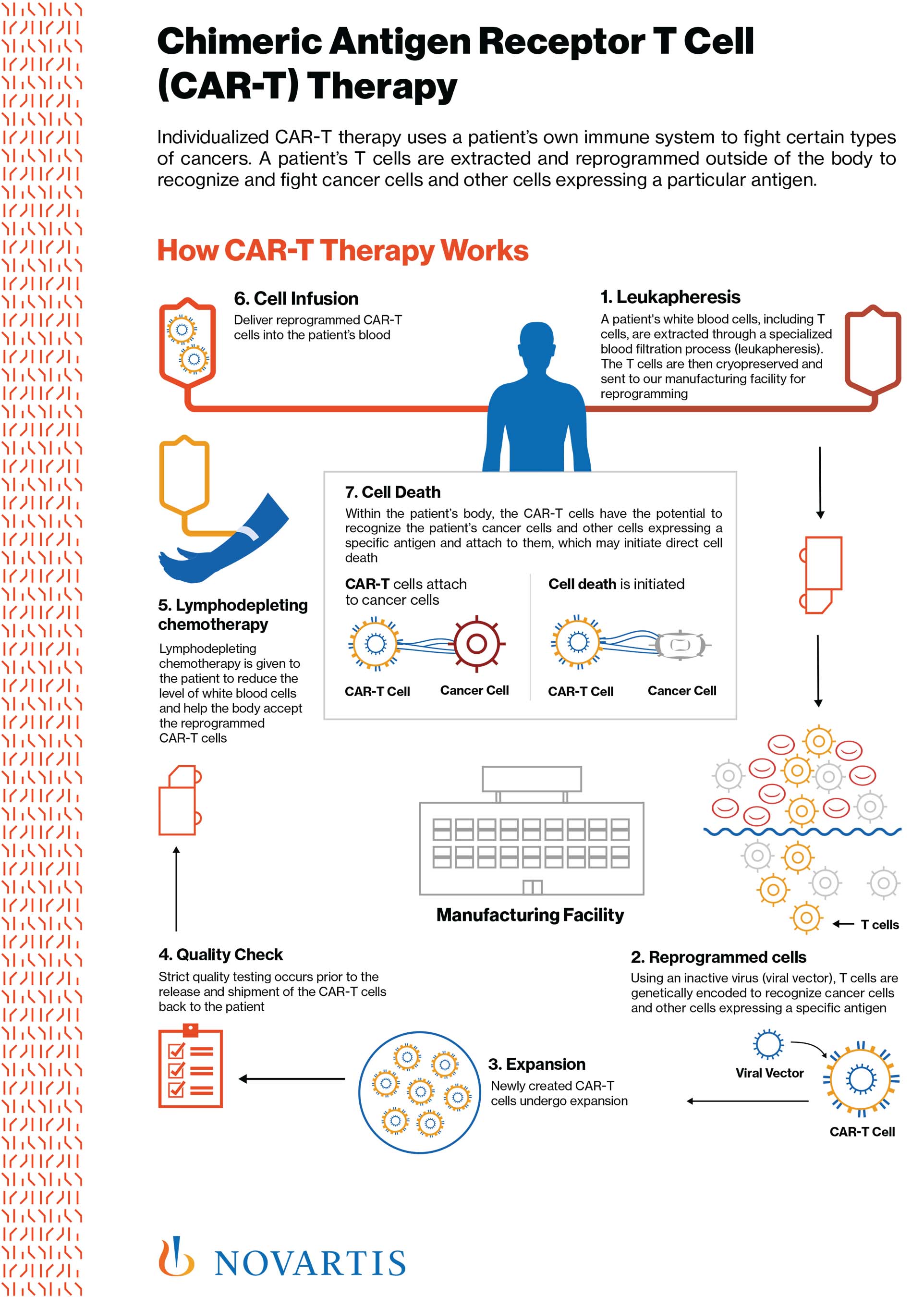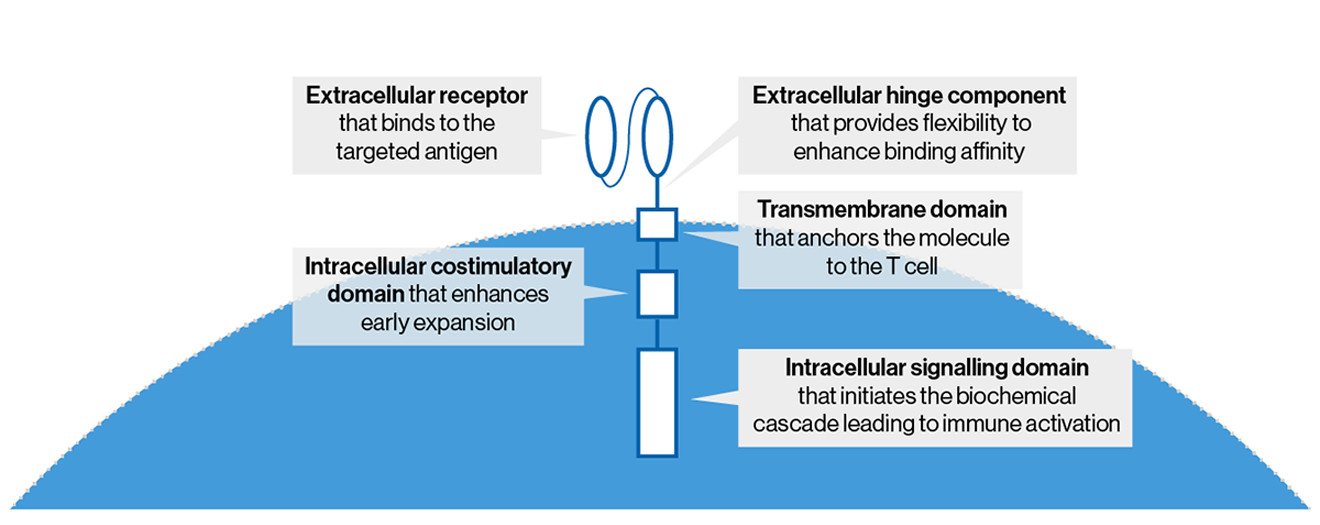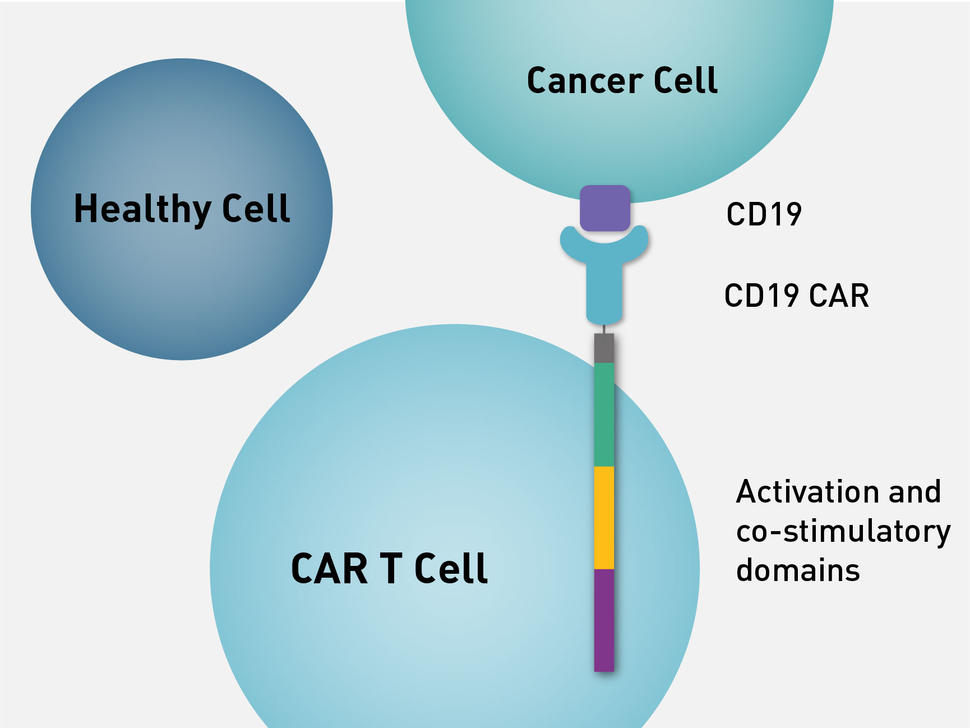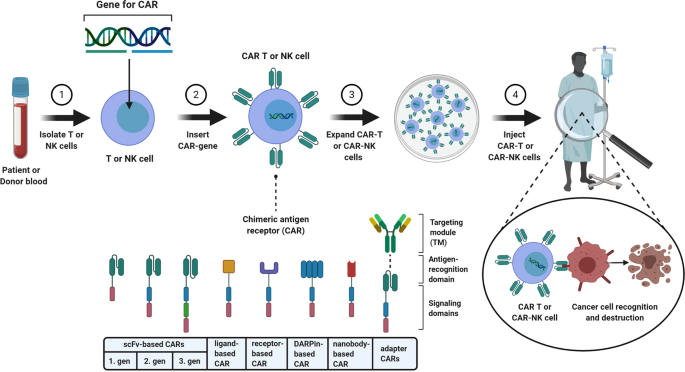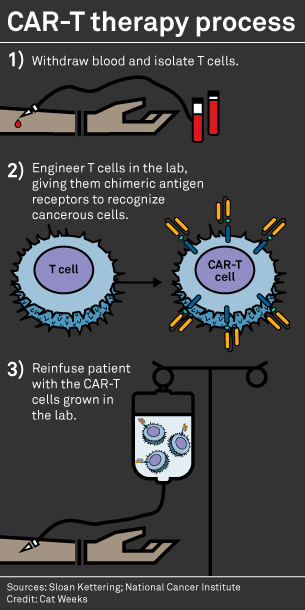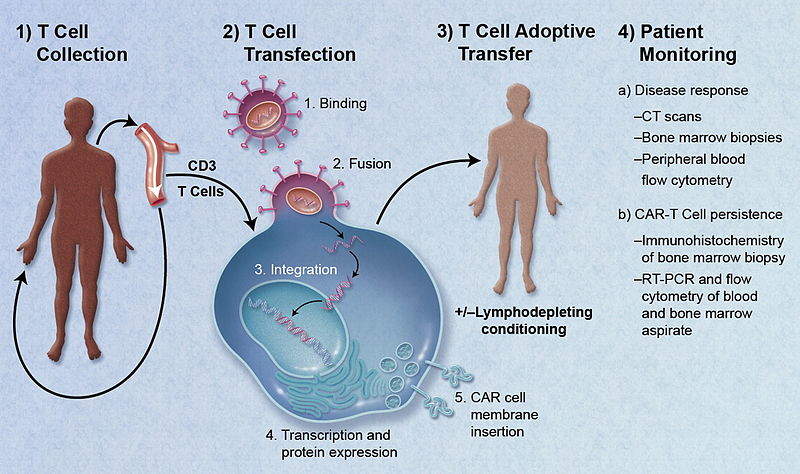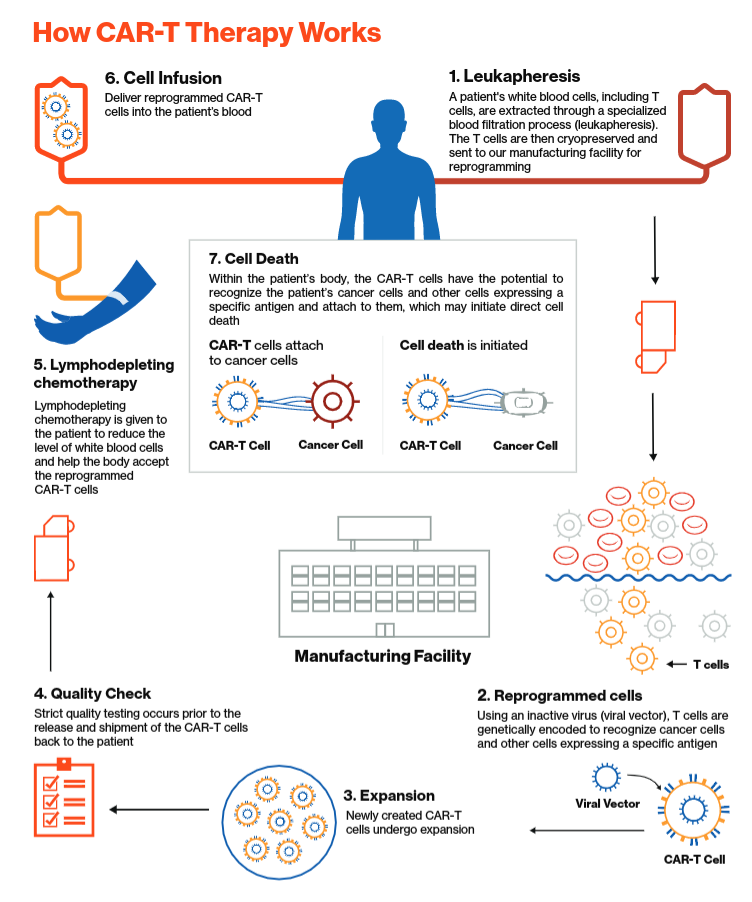
Anvisa aprova produto de terapia avançada para tratamento de câncer - ISMEP - Instituto Santa Marta de Ensino e Pesquisa
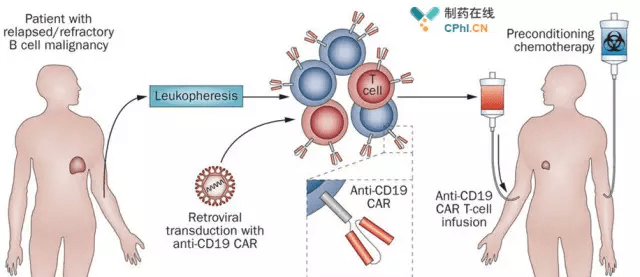
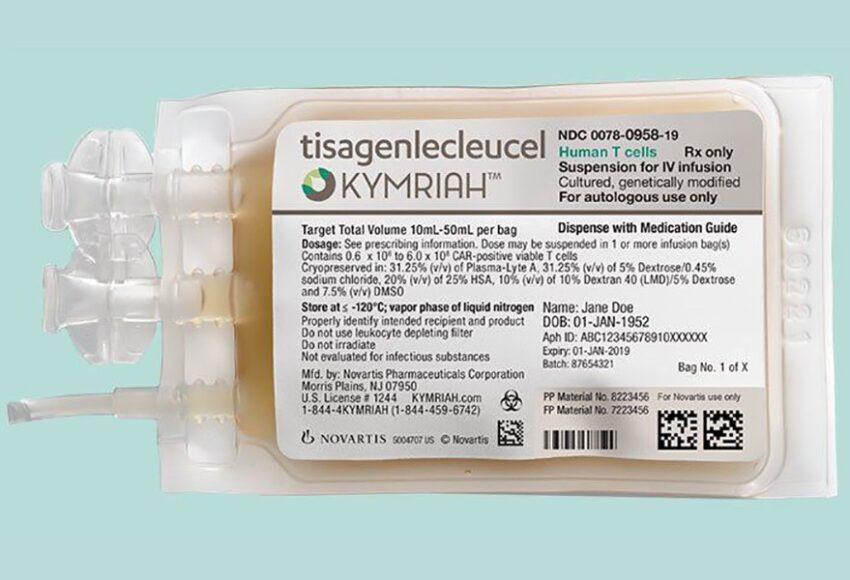
Historical moment! Milestone in immunotherapy: the world's first CAR-T cell product, CTL019, is approved for sale - Yongtai